Email Us
admin@axiomvetlab.co.uk
Oncology Service Now Available – FREE for 3 months
Hello Colleagues
Our ordering platform is now closed, our sister company MiLab Diagnostics are now handling our consumable orders.
If you have not received an invite for their platform please visit milab.store and submit a request, please ensure you populate all the fields on the request form.
Thank you, Team Axiom.

The skin is the largest organ of the body and one of the most frequently sampled tissues in veterinary diagnostic practice. As highlighted in Finn Pathologists’ Organ of the Month: Skin article (https://www.finnpathologists.com/2025/05/12/organ-of-the-month-skin/), skin biopsies remain central to the diagnosis of inflammatory, infectious, and neoplastic dermatoses. However, fine‑needle aspiration (FNA) cytology provides a complementary—and often underutilised—diagnostic approach, offering rapid, clinically actionable information before a biopsy is undertaken.
Because cutaneous and subcutaneous lesions are typically superficial, visible, and easily accessible, they are ideally suited to cytologic sampling. When performed correctly and interpreted within appropriate clinical context, FNA can reliably distinguish inflammatory from neoplastic disease, identify broad tumour lineage, and guide diagnostic and therapeutic planning while minimising patient morbidity.
The sample should ALWAYS be accompanied by a clinical history, as cytology lacks the architectural context provided by histopathology, and the aspirate may have been obtained from virtually any site on the animal.
Fine‑needle aspiration does not replace histopathology; rather, it guides and complements it. In day‑to‑day practice, FNA serves as a rapid screening and triage tool, helping clinicians decide:
FNA is particularly valuable as:
In many cases, FNA provides sufficient information to classify lesions into one of four broad categories: inflammatory, hyperplastic/reactive, benign neoplastic, or malignant neoplastic—a distinction that can dramatically influence case management.
Recommended technical considerations include:
Multiple needle passes from different parts of a lesion improve diagnostic yield and reduce sampling bias, especially in heterogeneous, cystic, or partially necrotic masses.
Material should be expelled gently onto clean slides and spread using squash, pull‑apart, or starfish techniques, depending on cellularity. Multiple smears should be prepared whenever possible.
To optimise diagnostic quality and reduce artefacts:
Suboptimal smear preparation remains one of the leading causes of non-diagnostic cytology, often more limiting than sampling technique itself.

Figure 1. A very thick FNA smear. The preparation is too dense to allow reliable identification of individual cells.

Figure 2. A very thin FNA smear. Cellular yield is low, but the presence of surface lipid is noteworthy, as this may indicate a lipoma. Such smears may not fix well after air‑drying, and cells can be lost during staining.
Artefact Examples
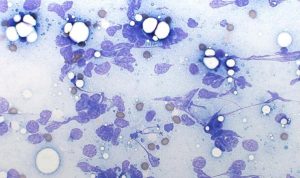
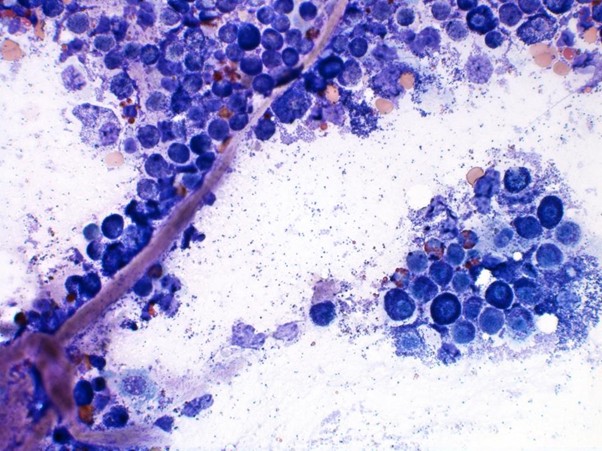
Figure 3. Excessive aspiration pressure causing widespread cell rupture (high‑grade lymphoma; Modified Wright’s, ×60).
Figure 4. Smear distortion and “rolled” material caused by spreading partially dried samples (Modified Wright’s, ×40).

Figure 5. Haemolysis artefact—serum proteins or haemoglobin precipitating along the smear during forceful aspiration (Modified Wright’s, ×60).
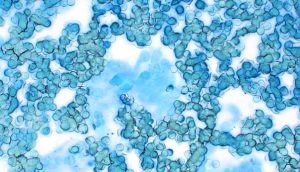
Figure 6. Formalin‑fume artefact resulting from transport too near biopsy pots, leading to poor staining and fixation changes (Modified Wright’s, ×60).
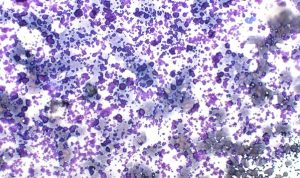
Figure 7. Ultrasound or lubricating gel contamination staining bright red /purple and obscuring detail (Modified Wright’s, ×40).
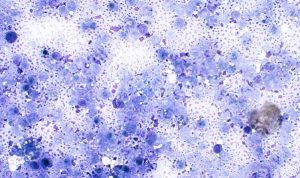
Figure 8. Necrotic material sampled from the centre of a lesion with few viable cells (Modified Wright’s, ×60)
Lesions and their underlying aetiology can be broadly subclassified according to the predominant cell populations harvested during sampling. Recognising these cytologic patterns (Figure 9) —whether inflammatory, round‑cell, epithelial, mesenchymal, or mixed—provides an immediate framework for generating meaningful differentials before more definitive testing.
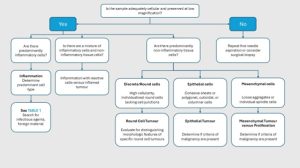
Figure 9. Schematic overview of the decision‑making algorithm employed during cytological evaluation.
Inflammatory lesions (and neoplasms) are among the most diagnostically rewarding samples in cutaneous cytology. Identifying the predominant inflammatory cell type helps guide the clinician toward likely underlying causes (Table 1).
Suppurative inflammation (Figure 10) is the pattern most frequently encountered. It is dominated by neutrophils—often degenerate—set within a proteinaceous or necrotic background. Intracellular bacteria support a diagnosis of sepsis, although extracellular organisms may also be present.
Other inflammatory patterns also provide important diagnostic clues:
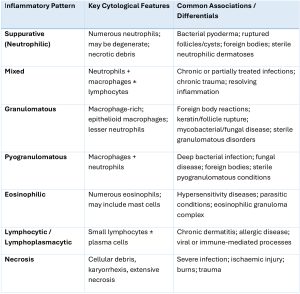
Table 1. Types of inflammatory patterns and their associated clinic lesions.
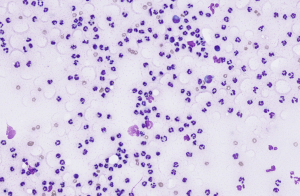
Round Cell Tumours
Round cell tumours exfoliate readily and typically provide highly cellular, diagnostically rewarding aspirates. They contain discrete, non-cohesive cells, making them particularly amenable to FNA evaluation.
Mast cell tumours (Figure 11) consist of round, discrete cells with abundant cytoplasmic metachromatic granules. Granule density may vary with tumour grade. Eosinophils are commonly present owing to mast cell–derived chemotactic mediators.
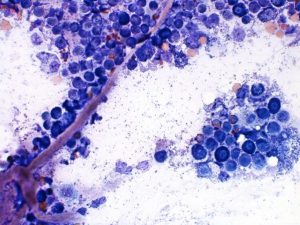
Figure 11 – Mast cells with obvious magenta granules and some capillaries. Modified Wright’s stain, ×60 objective.
Cutaneous histiocytomas (Figure 12) form sheets of histiocytic cells with pale to lightly basophilic cytoplasm and round to indented nuclei. Regressing lesions often contain numerous small lymphocytes, reflecting immune-mediated regression.
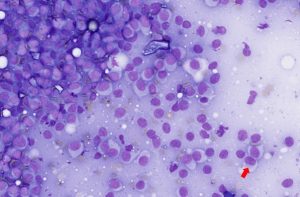
Figure 12 – Cytological features of a histiocytoma. The smear shows a population of medium‑sized round cells with abundant pale blue cytoplasm and a few reniform to indented nuclei (Red arrow), typical of reactive Langerhans‑cell origin. Modified Wright’s stain, ×40 objective.
Other, less common, round cell tumours in the skin include lymphoma, histiocytic sarcoma, and extramedullary plasma cell tumours.
Epithelial Tumours
Epithelial neoplasms usually exfoliate in cohesive clusters, sheets, or acinar arrangements due to strong intercellular adhesion.
Sebaceous adenomas (and more commonly, nodular areas of sebaceous hyperplasia) and sebaceous epitheliomas yield cohesive groups of epithelial cells with abundant foamy, lipid-rich cytoplasm and small, uniform nuclei. The presence of mature sebocytes with minimal pleomorphism supports a benign process; however, increased numbers of smaller basal cells raise suspicion for epithelioma (Figure 13).
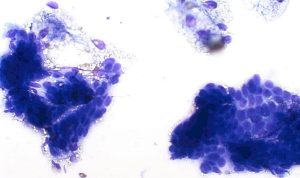
Figure 13 – Cytological features of a sebaceous epithelioma. The smear shows clusters and sheets of basaloid epithelial cells with some having abundant finely vacuolated, foamy cytoplasm consistent with sebaceous differentiation. Nuclei are round to oval with smooth chromatin and small nucleoli. Modified Wright’s stain, ×40 objective.
Cystic and keratinising lesions—including non‑neoplastic cysts—may yield only keratinaceous debris with scant or absent epithelial cells (Figure 14).
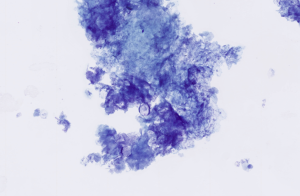
Figure 14 – Cytological features of keratinaceous debris from an epidermal cyst. The smear shows abundant anucleate keratin squames admixed with amorphous keratinous material. Modified Wright’s stain, ×40 objective.
Mesenchymal Tumours
Mesenchymal tumours exfoliate poorly and often produce low-cellularity preparations. When present, cells often appear spindle-shaped, stellate, or elongate, with wispy cytoplasm and oval to elongated nuclei. They frequently lie within or adjacent to pale extracellular matrix, supporting a mesenchymal origin (Figure 16).
Lipomas, a very common mesenchymal tumour, but these are not spindle-shaped, and are easily identified due to the presence of free lipid droplets and adipocytes distended with clear cytoplasm and a small peripheral nucleus (Figure 15).
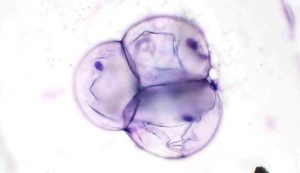
Figure 15 – Cytological features of a lipoma. The smear shows numerous mature adipocytes appearing as large round cells with clear, well‑demarcated cytoplasmic vacuoles causing marked peripheral displacement of small, dense nuclei. Modified Wright’s stain, ×40 objective.
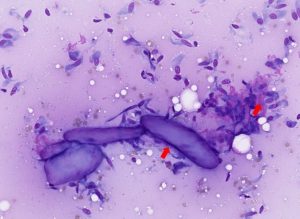
Figure 16 – Cytological features of a well‑differentiated sarcoma. The smear shows a population of spindle‑shaped mesenchymal cells arranged individually and in loose aggregates, with moderate pale cytoplasm and elongate to oval nuclei exhibiting mild anisokaryosis. Scattered extracellular matrix (collagen) may be present (red arrows). Modified Wright’s stain, ×40 objective
While cytology can strongly suggest a spindle cell tumour, differentiating benign from malignant is often unreliable, and histopathology is usually required for definitive classification.
Melanocytic Tumours
Melanocytic tumours show considerable cytologic variability. Melanin pigment may be abundant, scant, or absent (amelanotic forms), the latter making diagnosis more challenging.
Benign lesions (e.g., melanocytomas) typically contain well‑differentiated cells with minimal atypia. Malignant melanomas (Figure 17) often demonstrate marked anisocytosis and anisokaryosis, prominent nucleoli, and occasional bi‑ or multinucleation. Dense pigment granules—when present—may obscure nuclear detail. However, histopathology is required to make the distinction.
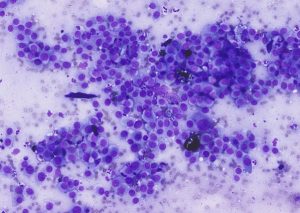
Figure 17 – Cytological features of a poorly differentiated melanoma. The smear shows a population of pleomorphic round to spindle‑shaped cells with scant to moderate basophilic cytoplasm and marked anisokaryosis. Nuclei are irregular with coarse chromatin and prominent nucleoli, and occasional binucleated or multinucleated forms may be present. Fine intracytoplasmic melanin pigment may be seen but is often sparse. Modified Wright’s stain, ×40 objective.
Common pitfalls include sampling error, overinterpretation of inflammation, failure to recognise poorly differentiated or low‑grade neoplasms, and suboptimal smear preparation. Awareness of these limitations—and clear communication of any diagnostic uncertainty—are essential to good clinical practice.
Fine needle aspiration cytology is a cornerstone of dermatological diagnosis. With careful technique and thoughtful pattern recognition, it provides rapid, valuable insights that support clinical decision‑making. Used in conjunction with histopathology, cytology enhances diagnostic efficiency, reduces unnecessary surgery, and improves patient outcomes.
Histopathology remains essential for definitive tumour classification, grading, margin assessment, and evaluation of complex or deep inflammatory dermatoses. Cytology is most powerful when used early in the diagnostic pathway, guiding biopsy site selection, assessing urgency, and informing clinical planning.
Muscle I. Muscle biopsy In this blog, we turn to muscle biopsies, specifically skeletal muscle. …
Mammary Glands I. Indications for biopsy/pathological evaluation Mammary lesions are among the most common samples…
PART I – The Male Reproductive Tract I. Indications for biopsy/pathological evaluation The male reproductive…
Organ of the month: Oral I. Indications for for oral biopsy Oral biopsies are among…
For receiving our news and updates in your inbox directly.

Manor House, Brunel Road, Newton Abbot, Devon, TQ12 4PB

The great explorer of the truth, the master-builder of human happiness no one rejects dislikes avoids pleasure itself because it is pleasure but because know who do not those how to pursue pleasures rationally encounter consequences that are extremely painful desires to obtain.
Read More



